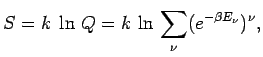Empiric entropy estimation
Since the interaction between hydrogen and PAHs is found to be very weak, 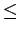 10 kJ
10 kJ mol
mol , it is expected that temperature increase will greatly influence the adsorption.
Thus, as first approximation experimental data is used to assess the entropy contribution.
In the gas phase it is governed by the five lowest frequencies of the H2-graphene system, corresponding to two rotations and three translations of H2.
Thermodynamic data calculated within the harmonic approximation are not expected to be reasonable as for these weak, low-frequency modes (they are found between 70 and 160 cm
, it is expected that temperature increase will greatly influence the adsorption.
Thus, as first approximation experimental data is used to assess the entropy contribution.
In the gas phase it is governed by the five lowest frequencies of the H2-graphene system, corresponding to two rotations and three translations of H2.
Thermodynamic data calculated within the harmonic approximation are not expected to be reasonable as for these weak, low-frequency modes (they are found between 70 and 160 cm ).
However, the low-frequency modes show that H2 will rotate nearly freely, which gives the appearance of a physisorbed sphere rather than a linear molecule.
Therefore, the physisorption cannot be compared only with the most stable orientation of H2 normal to the PAH, but has to be averaged for all orientations.
The harmonic approximation is not sufficient for the description of the thermodynamical functions of state for a weakly bound molecules.
Thus, the entropy of H2 physisorbed on graphite surface is estimated from experimental data:
The first vibrational levels of the laterally averaged H2 mode normal to graphite surface have been measured by Mattera et al. at low pressure [56].
From the first vibrational level one can obtain the zero-point energy (
).
However, the low-frequency modes show that H2 will rotate nearly freely, which gives the appearance of a physisorbed sphere rather than a linear molecule.
Therefore, the physisorption cannot be compared only with the most stable orientation of H2 normal to the PAH, but has to be averaged for all orientations.
The harmonic approximation is not sufficient for the description of the thermodynamical functions of state for a weakly bound molecules.
Thus, the entropy of H2 physisorbed on graphite surface is estimated from experimental data:
The first vibrational levels of the laterally averaged H2 mode normal to graphite surface have been measured by Mattera et al. at low pressure [56].
From the first vibrational level one can obtain the zero-point energy (
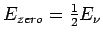 = 2.01 kJ
= 2.01 kJ mol
mol ).
The entropy contribution of the motion normal to the graphite surface can be estimated using partition function
).
The entropy contribution of the motion normal to the graphite surface can be estimated using partition function
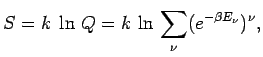 |
|
|
(2.3) |
with
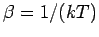 .
The correction to the zero point energy and entropy contributions is thus 2.3 kJ
.
The correction to the zero point energy and entropy contributions is thus 2.3 kJ mol
mol for temperature of liquid nitrogen (computed at 90 K), while at room temperature it is 3.4 kJ
for temperature of liquid nitrogen (computed at 90 K), while at room temperature it is 3.4 kJ mol
mol .
This correction is in the same order of magnitude as the physisorption energy of H2
.
This correction is in the same order of magnitude as the physisorption energy of H2 C6H6, but slightly smaller.
On the other hand the experimentally detected H2 physisorption energy on graphite surface at low temperatures (liquid nitrogen) is
C6H6, but slightly smaller.
On the other hand the experimentally detected H2 physisorption energy on graphite surface at low temperatures (liquid nitrogen) is 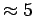 kJ
kJ mol
mol [56].
Given the value of
[56].
Given the value of 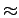 7 kJ
7 kJ mol
mol estimated by ab initio parametrised potential is thus supposed to be realistic.
Although this approximation suggests very small attractive interaction with the graphene surface, it is not enough to answer the question whether H2 will remain adsorbed, for example, inside porous carbon material.
estimated by ab initio parametrised potential is thus supposed to be realistic.
Although this approximation suggests very small attractive interaction with the graphene surface, it is not enough to answer the question whether H2 will remain adsorbed, for example, inside porous carbon material.
Lyuben Zhechkov
2007-09-04