One of the smallest fullerenes observed experimentally is C28[93].
In contrast to most fullerenes it has only one unique structure with tetrahedral symmetry 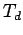 .[94]
The small size and four unpaired electrons in each set of four triplets of pentagons cause the high reactivity of the C28 cage. [95]
This fact was confirmed by more elaborate theoretical studies.[96,97]
The high reactivity, combined with the
.[94]
The small size and four unpaired electrons in each set of four triplets of pentagons cause the high reactivity of the C28 cage. [95]
This fact was confirmed by more elaborate theoretical studies.[96,97]
The high reactivity, combined with the 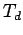 symmetry and the readiness to donate or to accept four electrons, opens the possibility to form solids based on tetrahedral C28 building blocks.
Numerous theoretical investigations report that the highly reactive C28 cage can be stabilised by an encapsulated metal atom.[97,98,99,100]
In particular, metal atoms M which readily form M^4+ ions (e.g., d-elements Ti, Zr, Mo, f-elements U, Pu, Ce, p-elements Si, Ge) are suitable to stabilise the tetravalent C28 cage.
An alternative route to saturate the free valences of the C28 radical is to form chemical bonds by linking with hydrogen, halogen atoms, alkyl groups or by the formation of polymers, films or crystals.[97,101,102]
The four unpaired electrons localised at the four pentagon triplets in a tetrahedral arrangement make the coordination of C28 fullerene cages similar to an
symmetry and the readiness to donate or to accept four electrons, opens the possibility to form solids based on tetrahedral C28 building blocks.
Numerous theoretical investigations report that the highly reactive C28 cage can be stabilised by an encapsulated metal atom.[97,98,99,100]
In particular, metal atoms M which readily form M^4+ ions (e.g., d-elements Ti, Zr, Mo, f-elements U, Pu, Ce, p-elements Si, Ge) are suitable to stabilise the tetravalent C28 cage.
An alternative route to saturate the free valences of the C28 radical is to form chemical bonds by linking with hydrogen, halogen atoms, alkyl groups or by the formation of polymers, films or crystals.[97,101,102]
The four unpaired electrons localised at the four pentagon triplets in a tetrahedral arrangement make the coordination of C28 fullerene cages similar to an 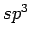 -hybridised carbon atom.
Two crystalline formations of
-hybridised carbon atom.
Two crystalline formations of 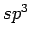 -carbon are found in nature: diamond and lonsdaleite (hexagonal diamond), suggesting that similar phases with C28 units instead of carbon atoms might exist.
-carbon are found in nature: diamond and lonsdaleite (hexagonal diamond), suggesting that similar phases with C28 units instead of carbon atoms might exist.
Crystallographically classified by the space groups Fd3m and P63/ mmc, these fulerites are known respectively as hyperdiamond (HD) and hyperlonsdaleite (HL) C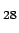 phases.
Shortly, they can be described as diamond and lonsdaleite crystals where the carbon atoms are substituted by the C
phases.
Shortly, they can be described as diamond and lonsdaleite crystals where the carbon atoms are substituted by the C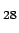 cages.
In both crystals, the intercage bonds are formed between the central atoms of the four C
cages.
In both crystals, the intercage bonds are formed between the central atoms of the four C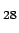 pentagon triplets which are present in each cage.
Due to the presence of large cavities between covalently bound C28 cages, fullerites are very lightweight materials with a mass density even lower than that of other graphite modifications (respectively 0.055 and 0.081 carbon atoms per Å
pentagon triplets which are present in each cage.
Due to the presence of large cavities between covalently bound C28 cages, fullerites are very lightweight materials with a mass density even lower than that of other graphite modifications (respectively 0.055 and 0.081 carbon atoms per Å ).
The 3D interaction potentials of a H2 guest molecule with the hyperdiamond/hyperlonsdaleite host structures are given in Fig. 6.4.
).
The 3D interaction potentials of a H2 guest molecule with the hyperdiamond/hyperlonsdaleite host structures are given in Fig. 6.4.
Figure:
H2 interaction potential in hyperdiamond (Left) and
hyperlonsdaleite (Right) like C28
interaction potential in hyperdiamond (Left) and
hyperlonsdaleite (Right) like C28 fullerites. The
colour of the iso-surfaces coresponds to the energy bar in kJ
fullerites. The
colour of the iso-surfaces coresponds to the energy bar in kJ mol
mol .
.
|
Both structures have tubular areas where the H2 interaction potential is attractive.
For HL, these areas are more pronounced.
The potential is only attractive in the cavities between the fullerene cages and reaches well depths of about 9.5 and 13.1 kJ mol
mol , respectively, for hyperdiamond and for hyperlonsdaleite.
These values are comparable with those of graphene bilayers or C60 intercalated graphite (see Fig. 3.1 and 4.1).
The volume possessing only the attractive part of the potential for both host structures is integrated to be about 30 %.
This is 10 % lower than for the same volume confined between graphene layers (Chapter 3), but almost twice as bigger than C60 intercalated graphite (Chapter 4) and the modified graphite (section 6.1).
However, despite the large attractive area the entropy still has large contributions to the interaction free energy if the temperature is increased.
Thus, the H2 interaction free energy results to
, respectively, for hyperdiamond and for hyperlonsdaleite.
These values are comparable with those of graphene bilayers or C60 intercalated graphite (see Fig. 3.1 and 4.1).
The volume possessing only the attractive part of the potential for both host structures is integrated to be about 30 %.
This is 10 % lower than for the same volume confined between graphene layers (Chapter 3), but almost twice as bigger than C60 intercalated graphite (Chapter 4) and the modified graphite (section 6.1).
However, despite the large attractive area the entropy still has large contributions to the interaction free energy if the temperature is increased.
Thus, the H2 interaction free energy results to 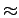 4.5 kJ
4.5 kJ mol
mol , twice smaller than in porous graphite (
, twice smaller than in porous graphite (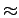 8.5 kJ
8.5 kJ mol
mol ), but about the same compared with C60 intercalated graphite (
), but about the same compared with C60 intercalated graphite (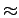 5 kJ
5 kJ mol
mol ).
In other words, the host model structures will attract hydrogen molecules much weaker.
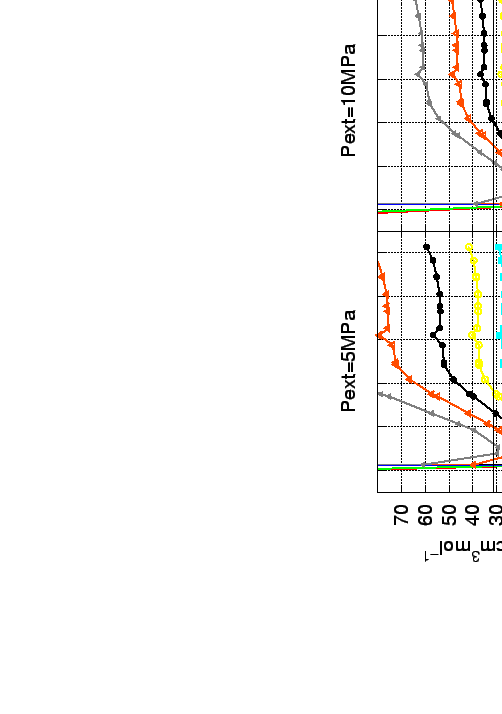
A direct evidence is the volumetric capacities of the system shown on Fig. 6.5.
).
In other words, the host model structures will attract hydrogen molecules much weaker.
A direct evidence is the volumetric capacities of the system shown on Fig. 6.5.
Figure:
Volumetric (Top) and gravimetric (Bottom) storage capacities for solid C28
diamond and lonsdeleit fullerites, given for various temperatures (see the legend) and pressures.
The symbols on the curves denote that the approximation is within the limits (pressure and temperature) of the real gas
equation of state [36]. The targets[4] for automotive
applications (Gwt) 6.0 %,(V) 44.4 cm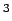 /mol) are indicated as horizontal lines.
/mol) are indicated as horizontal lines.
 |
The decrease of the hydrogen density inside HL and HD cavities compared with other models (see Fig. 5.7, 5.12, 6.3) is closely related to the cavity's special features.
In contrast to nanotubes, carbon foams and porous graphite, where the H2 attractive force is due to the 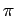 overlapping inside the cavities, the contribution to HL and HD interaction potentials comes only from the outer surfaces of C28 cages.
This can be easily understood if one considers for example an interaction potential inside and outside of a C60 fullerene cage.
While inside the cage all carbon atoms contribute equally to the H2
overlapping inside the cavities, the contribution to HL and HD interaction potentials comes only from the outer surfaces of C28 cages.
This can be easily understood if one considers for example an interaction potential inside and outside of a C60 fullerene cage.
While inside the cage all carbon atoms contribute equally to the H2 fullerene interaction, outside of the cage
fullerene interaction, outside of the cage given the
given the 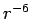 relation
relation the main contribution would come only from the nearest atoms.
Hence, the negatively bent surfaces are less effective for trapping the hydrogen molecules.
On the other hand, due to their low carbon density HL and HD gravimetric capacities are lower by less than 1 gwt % from the previously mentioned materials.
Since the hydrogen density decreases, the energy density is going to be insufficient as well.
Therefore, high surface area materials with high gravimetric storage capacities should be obligatory discussed together with their volumetric capacities in order to avoid ambiguities.
Lyuben Zhechkov
2007-09-04
the main contribution would come only from the nearest atoms.
Hence, the negatively bent surfaces are less effective for trapping the hydrogen molecules.
On the other hand, due to their low carbon density HL and HD gravimetric capacities are lower by less than 1 gwt % from the previously mentioned materials.
Since the hydrogen density decreases, the energy density is going to be insufficient as well.
Therefore, high surface area materials with high gravimetric storage capacities should be obligatory discussed together with their volumetric capacities in order to avoid ambiguities.
Lyuben Zhechkov
2007-09-04
![]() phases.
Shortly, they can be described as diamond and lonsdaleite crystals where the carbon atoms are substituted by the C
phases.
Shortly, they can be described as diamond and lonsdaleite crystals where the carbon atoms are substituted by the C![]() cages.
In both crystals, the intercage bonds are formed between the central atoms of the four C
cages.
In both crystals, the intercage bonds are formed between the central atoms of the four C![]() pentagon triplets which are present in each cage.
Due to the presence of large cavities between covalently bound C28 cages, fullerites are very lightweight materials with a mass density even lower than that of other graphite modifications (respectively 0.055 and 0.081 carbon atoms per Å
pentagon triplets which are present in each cage.
Due to the presence of large cavities between covalently bound C28 cages, fullerites are very lightweight materials with a mass density even lower than that of other graphite modifications (respectively 0.055 and 0.081 carbon atoms per Å![]() ).
The 3D interaction potentials of a H2 guest molecule with the hyperdiamond/hyperlonsdaleite host structures are given in Fig. 6.4.
).
The 3D interaction potentials of a H2 guest molecule with the hyperdiamond/hyperlonsdaleite host structures are given in Fig. 6.4.
